Our BusinessRegenerative Medicine Business

Creating the future with regenerative medicine
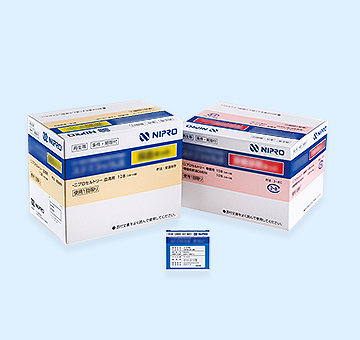
We contribute to the development of regenerative medicine, including autologous bone-marrow mesenchymal stem cells. This is the world’s first regenerative medicine product that has been approved for manufacturing and marketing (with conditions and time limits) for use in the treatment of patients with traumatic spinal cord injury.
The products on the English website may not be available locally. Detail of intended purpose may also vary by local.
Please contact with our local representative in more detail.